Research illuminates earliest part of phase separation
Scientists at St. Jude Children's Research Hospital are studying liquid-liquid phase separation (LLPS), a biophysical process through which proteins and nucleic acids in a cell are compartmentalized without a membrane. The work provides new insight into how the strength of the forces that drive phase separation are linked to the speed at which it occurs. The findings were published today in Nature Communications.
Cells need to sort and organize proteins and other components. One way they do so is through LLPS, a process similar to the way oil forms droplets in water. Membraneless organelles, bodies in a cell that behave like liquid droplets, organize certain proteins without enclosing them with a membrane. Instead, the proteins are held together by the biophysical forces that drive LLPS.
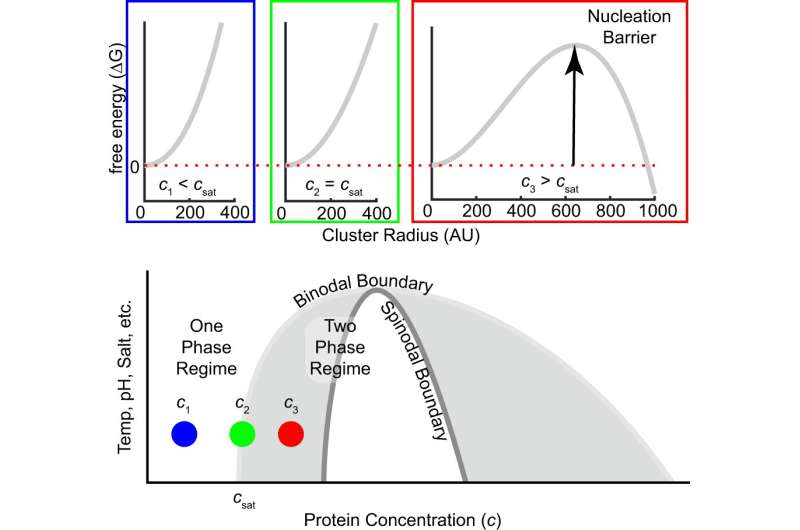
Little is known about how biomolecules transition from a one-phase solution to a two-phase mixture, in a process called nucleation. This process has been challenging to evaluate because it requires looking at extremely fast time scales (microseconds to milliseconds). As a result, most studies have looked instead at evolution within systems that are already in the two-phase regime (how droplets fuse and grow).
"We can see during these very early time points that even individual disordered protein molecules that phase separate have quite different properties than disordered proteins that don't phase separate," said co-corresponding author Tanja Mittag, Ph.D., St. Jude Department of Structural Biology.
Research at St. Jude and elsewhere has shown that LLPS can be involved in neurodegenerative diseases such as amyotrophic lateral sclerosis (ALS) and cancer. A better understanding of LLPS may ultimately lead to opportunities to therapeutically target parts of the process.
Microsecond time measurements give new understanding
The prevailing view of LLPS in biology has been that a change in cellular conditions can spontaneously lead to nucleation, switching a single-phase solution into two phases. In this research, the scientists used a simplified system, with a single protein in water and salt, to study whether the switch actually occurred in such a manner or if additional steps are required to kickstart the process.
With a technique called rapid mixing, time-resolved, small-angle X-ray scattering (TR-SAXS), the researchers were able to observe the earliest stages of the process. They examined the nucleation of a prion-like domain called A1-LCD from the protein hnRNPA1. Mutations of this protein cause ALS and other diseases.
The scientists showed how clusters of A1-LCD form, and how these clusters lead to LLPS. The findings indicate that nucleation contains distinct steps, differentiated by the size of the cluster. When looking at the smallest clusters (with few individual molecules), scientists found that the assembly behavior differed from classical nucleation theory. These deviations potentially explain why phase separation of some biomolecules can happen in milliseconds while it takes hours for others.
"Even in a simplified system, you still have to take into account these kind of non-ideal effects at the early stages of nucleation," said co-corresponding author Erik Martin, Ph.D., St. Jude Department of Structural Biology. "Before you can start thinking about phase separation assembly or condensation in cells, you need to think about things from a molecular level. There are going to be initial steps to that assembly that aren't accounted for in previous models."
More information: Erik W. Martin et al, A multi-step nucleation process determines the kinetics of prion-like domain phase separation, Nature Communications (2021).
Journal information: Nature Communications
Provided by St. Jude Children's Research Hospital