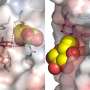
An illustration of the density of a water molecule network (blue surface map and red spheres) near the catalytic site (green sticks), projected on the surface of a plant exo-hydrolase, coloured by electrostatic potential (white and red surface maps). Credit: Maria Hrmova
Researchers from the University of Adelaide have discovered how water molecules are organized during plant hydrolytic reactions, knowledge that could have sweeping consequences for the biomedical, pharmaceutical, food and chemical industries.
The research team, led by Professor Maria Hrmova from the University of Adelaide, identified enzyme components that underlie water molecule networks and function as principal operators to regulate water flux during hydrolytic reactions.
"Conceptually, one of the most exciting undertakings in biophysics and biochemistry is to investigate the dynamics of water molecules," said Professor Hrmova, whose study was in Communications Biology.
"Water molecules are tiny chemical entities that behave in such a way that one moment you can see them, and the next you cannot. In this work, our international team deployed enzyme kinetics, high-resolution X-ray synchrotron crystallography, advanced molecular dynamics and prediction tools, such as ancestral sequence reconstruction, to understand the roles of water molecules.
"This multidisciplinary approach allowed us to understand their evolutionary trajectories and formulate principles for water molecule dynamics in hydrolytic reactions and how water molecules form harmonized or non-random networks at atomic levels," added Professor Hrmova.
Water is one of the smallest and most abundant molecules in the universe. It fulfills multiple metabolic roles as a solvent, substrate, cofactor, intermediate, and product, during biochemical transformations in living systems, such as plants and animals.
There are up to 80,000 enzymes fundamental to life that use water as a reactant, catalyzing and speeding up biochemical reactions upon which almost all metabolic and physiological processes depend.
These processes include the hydrolysis of carbohydrate substrates such as cellulose, starch, and other glycosides during the growth and development of all forms of life. This function allows enzymes, including plant hydrolases, to efficiently recycle polymeric substrates and support primary root extension, seed germination, and pollination.
In addition to the multibillion-dollar biomedical, pharmaceutical, food and chemical industries, this discovery could impact enzyme design and bioengineering, food, paper, pulp, bioplastics and textile materials processing, and biofuel production.
"Discoveries such as these are significant for product manufacturing through biotechnologies and foster the development of novel bioengineered hydrolytic enzymes," Professor Hrmova said. "These optimized enzymes could also function outside biological systems to produce pharmaceuticals, nutra-chemicals, drugs, chemicals, herbicides, pesticides, and other reagents."
This study builds on prior foundational work by Professor Hrmova and her team in the School of Agriculture, Food and Wine and the Waite Research Institute.
"The interdisciplinarity of our work—integrating techniques, tools, concepts, and theories—has allowed us to resolve the enigma of these processes among complex research challenges," she explained.
"In a broader context, here and in other studies, we identified the operators regulating water flux and networks during hydrolytic reactions, which—with other central phenomena such as processivity and reactant movements through trajectories—are all fundamental to catalysis," said Professor Hrmova.
Animated visualizations describing the evolution of water networks in a plant exo-hydrolase are available .
More information: Sukanya Luang et al, The structure and dynamics of water molecule networks underlie catalytic efficiency in a glycoside exo-hydrolase, Communications Biology (2025).
Journal information: Communications Biology
Provided by University of Adelaide